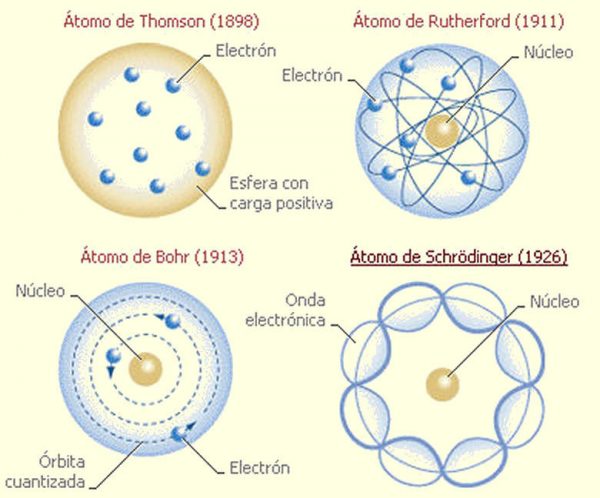
As in Bohr’s model, the energy of an electron in an atom is quantized it can have only certain allowed values. Each wavefunction is associated with a particular energy.The requirement that the waves must be in phase with one another to avoid cancellation and produce a standing wave results in a limited number of solutions (wavefunctions), each of which is specified by a set of numbers called quantum numbers. In January 1921, Schrdinger finished his first article on this subject, about the framework of the Bohr-Sommerfeld. Schrödinger incorporated Legendre’s equations into his wavefunctions. Schrdinger's first publications about atomic theory and the theory of spectra began to emerge only from the beginning of the 1920s, after his personal acquaintance with Sommerfeld and Wolfgang Pauli and his move to Germany. 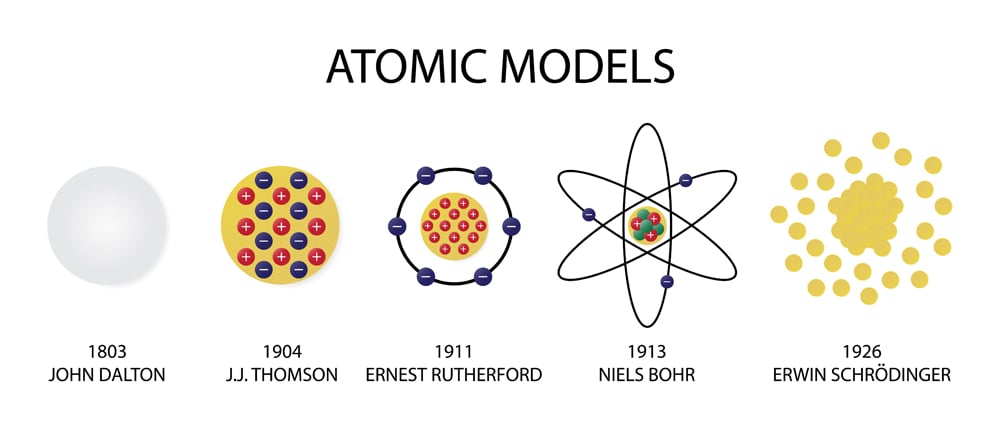
Fortunately, however, in the 18th century, a French mathematician, Adrien Legendre (1752–1783), developed a set of equations to describe the motion of tidal waves on the surface of a flooded planet. 
His atomic theory had five statements: British Chemist and Physicist, John Dalton (17661844. He was a schoolteacher who performed many experiments on atoms. The atom is now considered to be the basic unit of simple substances or elements. From the patterns of one- and two-dimensional standing waves shown previously, you might expect (correctly) that the patterns of three-dimensional standing waves would be complex. John Dalton (17661844), an English Chemist, put forward a theory to describe the nature of the atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
